
John Dalton believed that elements are composed of indivisible atom. There is a callout which shows that copper is made up of many sphere-shaped atoms. This chemistry video tutorial provides a basic introduction into Dalton's Atomic Theory. The left stoppered bottle contains copper and oxygen. (credit copper: modification of work by Copper ). It was 1913 before English physicist Henry Moseley reorganized the periodic table by atomic number.\): When the elements copper (a shiny, red-brown solid, shown here as brown spheres) and oxygen (a clear and colorless gas, shown here as red spheres) react, their atoms rearrange to form a compound containing copper and oxygen (a powdery, black solid). 
He thought it was a question of inaccurate measurement or other experimental error. When he flipped his chart to a horizontal table two years later, he created a form much like what you see in chemistry textbooks and on the walls of chem labs today.Īlas, Mendeleev's table was based on atomic mass rather than atomic number, so details like the placement of tellurium and iodine didn't work out. The simplified notation led the way for English analytical chemist John Newlands to formulate his Law of Octaves and a prototype periodic table of the elements in 1864, but it was Russian chemist Dmitri Mendeleev who really laid it all on the table with 63 elements in 1869. Any pure compound will always have the same ratio of the same elements. Atoms of different elements vary in size, mass, and chemical behavior. All atoms of the same element are identical and have the same mass. 
The five main points are: Matter is made up of atoms, small and indivisible particles. 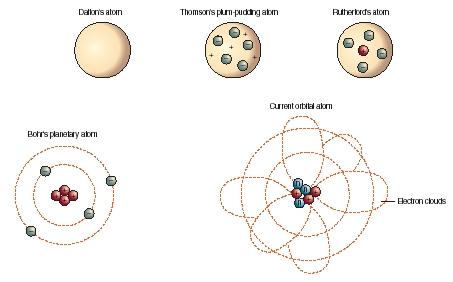
So it's Au for gold and Ag for silver, not the circled G and S of Dalton's original notation. This is the basic idea of the law of constant composition. Daltons Atomic Theory was formulated by John Dalton in 1808, and it remains a fundamental tenet of chemistry to this day. In an era when all Europe's learned men (and the few women who were allowed into schools and universities) knew Latin, the shared language was an international lingua franca.Īll but a handful of Berzelius' symbols are still used today. Berzelius organized 47 elements with letters alone, and he based those letters not primarily on the English names, but on the Latin ones. Half of Dalton's symbols used letters inside a circle to represent the element. He also researched colour blindness, which he had as a result, colour blindness is known as Daltonism in several languages. Composition: Democritus believed that atoms were indivisible, while Dalton proposed that atoms had a fixed atomic weight and could be broken down into smaller particles. That all atoms of a specific element have a characteristic mass. He introduced the atomic theory into chemistry. Origin: Democritus’ atomic theory was based on philosophical reasoning, while Dalton’s atomic theory was based on empirical evidence.Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4 ), is a fundamental concept that states that all elements are composed of atoms. That atoms have a definite physical reality (characteristic size) The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 2.1.4 2.1.Rather, I will focus on the key features of Dalton’s theory: 2 I won’t reiterate the four postulates here. A decade after Dalton formulated his symbols, Swedish chemist Jöns Jakob Berzelius simplified the system. We start with John Dalton’s atomic theory (published in stages from 1803 to 1810) and the billiard ball model of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
